Chris Anderson: Dr. Şahin and Dr. Türeci, welcome. Such a treat to speak with you.
克里斯·安德森: 吳沙忻博士,圖雷西博士, 歡迎,非常榮幸能與你們對談。
Özlem Türeci: Thank you very much, Chris. It's a pleasure to be here.
圖:非常謝謝你,克里斯。 很榮幸能參與。
CA: So tell me, as you think back over the last 18 months, what words pop to mind for you?
克:回想過去十八個月, 你的腦中最先浮現的是什麼詞句?
ÖT: Well, one word which comes to mind is breathless. It was indeed a breathless 16, 17 months for us. When we started in January last year, it was already at that time clear to us that we were already in a pandemic. What was not known was how fast this pandemic would evolve and whether we would have the time in the first place to have a vaccine ready soon enough in due time. And understanding this, it meant for us that there was not even one day to lose. And this was the mindset of the entire team here in Mainz and at BioNTech and later on also of our partners which were involved, Pfizer and others, to keep going and be fast.
圖:腦中最先浮現的是屏息以待。 對我們來說,這真的是 屏息以待的十六、十七個月。 去年一月,我們開始時, 當時我們就很清楚 我們已經在疫情當中了。 那時不知道的, 是疫情會演化得這麼快, 以及我們到底能不能 把疫苗準備好, 且要夠快,夠即時。 因為了解這一點, 這對我們來說意味著 我們連一天都不能損失。 整個團隊都抱持這種心態, 不論是在美茵茲這裡, 還是在 BioNTech 皆是如此。 還有後續參與的合作夥伴, 輝瑞及其他夥伴, 都想持續邁進且動作要快。
CA: I mean, it's so extraordinary that the ideas and the work in your minds have now impacted hundreds of millions, perhaps billions of people. That must feel overwhelming. And yet, I know at the same time, you don't believe in this notion of a flash-in-the-pan ideas. Steven Johnson, the author, in his book "Where [Good] Ideas Come From," speaks of the slow hunch, that the best ideas happen over many years. And I know that you believe that is true in your case. I'd like us to go back a couple of decades to -- tell us how this began. How did you meet?
克:這真的很不簡單, 就是你們的想法及工作 現在影響了數億人, 甚至數十億人。 這感覺一定很受寵若驚。 然而,同時我也知道, 這不是靈光閃現。 作家史蒂文·強森在他的書 《創意從何而來》中 談到緩慢的預感,就是最佳的點子 要花很多年才能產生。 我知道你們也如此認為。 我想倒帶數十年, 請你們談談這是如何開始的。 你們怎麼認識的?
ÖT: We met on an oncohematology ward, Uğur being a young physician, and I was still in medical school training on ward. Which means we met in one of the worlds which became important to us, the world of patient care, of treating oncohematology patients. And we soon found out that there was a second world which we liked, namely the world of science. We were haunted by the same dilemma, namely that whereas there was not much we could offer our cancer patients, there were so many potential technologies we encountered in the lab which could address this. So one of our shared visions was to bridge this dilemma by working on bringing science and technology fast. And that's an important word here. Fast to the patient's bedside to address high medical need.
圖:我們在血液腫瘤科病房認識, 沙忻當時是年輕的醫師, 我還在讀醫學院, 在病房接受訓練。 也就是說,讓我們相遇的世界, 後來變成對我們很重要的世界, 就是照護病人的世界, 治療血液腫瘤科的病人。 我們很快就發現, 還有一個我們都喜愛的世界, 也就是科學的世界。 我們被同樣的窘境所困擾, 也就是,雖然, 我們能提供給癌症病人的不多, 但在實驗室中, 我們發現好多有潛力的技術, 可能可以用來處理這個問題。 所以,我們共同的遠景之一, 是要橋接這種兩難, 要把科學與技術 快速連結。 在此,快速這個詞很重要。 快速將解方帶到病人的床邊, 以滿足高度的醫療需求。
CA: So I think the first company you founded nearly 20 years ago was to use the power of the human immune system to tackle cancer.
克:所以你們在近二十年前 創立的第一間公司 是在利用人類免疫系統的力量 來對付癌症。
Uğur Şahin: We were always interested in using the patient's immune system
吳:我們一直都很想
to fight cancer and other type of diseases. As immunologists, we knew how powerful the human immune system is. But it was also clear that the human immune system, in the case of cancer, did not fight cancer cells. It could fight it, but it didn't. And for that, we wanted to develop immunotherapies. That means treatments that use the power of the immune system and redirect the power of the immune system to cancer cells. It was clear that in the university setting, we could not continue to develop monoclonal antibodies because the cost for development of monoclonal antibodies before you can start a clinical trial, was in the range of 20, 30 million euros, and therefore we decided to start a company to get the funding.
用病人的免疫系統 來對抗癌症和其他疾病。 身為免疫學家, 我們知道人類的免疫系統有多強大。 但,我們也很清楚, 人類免疫系統 在遇到癌症時, 不會對抗癌細胞。 它可以對抗,但卻沒有。 因此, 我們想要發展免疫治療。 也就是, 用免疫系統的力量來做治療, 將免疫系統的力量重新導向癌細胞。 很明顯,在大學的環境中, 我們無法繼續發展單株抗體, 因為發展單株抗體的成本, 在還沒有開始臨床實驗之前 就已經要花到兩、三千萬歐元, 因此我們決定創立 一間公司來取得資金。
CA: Now, soon after you started this company, you decided to get married. Tell me about your wedding day.
克:你們成立這間公司後 沒多久,就決定結婚了。 能否談談你們的結婚日。
ÖT: Day was well planned, a quick wedding. And thereafter we went back to the laboratory and our guests at our wedding, that was basically our team, our research team. So no time to lose, Chris.
圖:那天的規劃很好, 快速的婚禮。 結束後我們就回到實驗室, 婚禮的賓客基本上都是我們的團隊, 我們的研究團隊。 分秒必爭,克里斯。 克:(笑)
CA: (Laughs) That was a pretty special honeymoon. I mean, it seems like your love for each other is very much bound up in your love for this work and your sense of the importance of this work. How would you characterize those intersecting relationships there?
挺特別的蜜月。 你們對彼此的愛似乎 緊繫著你們對這份工作的愛, 以及你們對這份工作的重視。 你們會怎麼描述那些交錯的關係?
UŞ: We are really two scientists. At the end of the day, we love what we do, and for us, we don't differentiate between work and life balance. It's for us really a privilege to be scientists, to be able to do what we love. And therefore, we combine our normal life with our professional life. And therefore, this is pretty normal for us.
吳:我們其實就是兩個科學家。 到頭來,我們熱愛我們的工作, 而且,對我們而言,我們不用 去區別工作和生活的平衡。 我們覺得三生有幸能夠成為科學家, 能夠去做我們熱愛的事。 因此, 我們把日常生活和工作生活結合。 因此,這對我們而言挺正常的。
CA: So talk to me about this extraordinary molecule RNA, and how you got interested in it and how it became, as I understand it, an increasing focus of your work. And indeed, it led to the founding of BioNTech. Talk about that.
克:能否談談這個 不可思議的分子 RNA, 你們怎麼會對它產生興趣? 還有,就我的了解, 它變成你們工作的重心? 它也的確為 BioNTech 帶來了資金。 跟我們談談這些。
UŞ: Yeah, mRNA is a natural molecule, it's one of the first molecules of life. It is a carrier of genetic information. But in contrast to DNA, it's not stable. So it can be used to transfer information to human cells. And the human cells can use this information to build proteins, which can be used for therapeutic settings, for example, to make a protein which is a vaccine, or to make a protein which is an antibody, or to make a protein which is another type of drug. And we were fascinated by this molecule class, because it was very clear that mRNA can be produced pretty fast, within a few days. And we were, as MDs, we were particularly interested to develop personalized medicines. That means a treatment and immunotherapy specifically designed for a cancer patient, because one of the key challenges in cancer treatment, is that every patient has a different tumor. If you compare two tumors of two patients with the same type of tumor, the similarity of the tumors is less than three percent and 97 percent is really unique. And today, it's still not possible to address the uniqueness of the tumor of a patient. And therefore, we were seeking for a technology which could be used for immunotherapy and which could be used to develop a treatment within the shortest possible time. The idea to get the genetic sequence of the tumor and then make a vaccine which is personalized, within a few weeks.
吳:好的,mRNA 是種天然分子, 它是生命最早的分子之一。 它帶著遺傳訊息。 但和 DNA 相反,它並不穩定。 所以可以用它 傳遞訊息給人類細胞。 人類細胞 則可以利用這些訊息來創造蛋白質, 然後拿來治療, 比如, 製造當作疫苗用的蛋白質, 或製造當作抗體用的蛋白質, 或製造當作其他藥物用的蛋白質。 我們對這類分子十分著迷, 因為, 我們非常清楚, 製造 mRNA 的速度很快, 只需要幾天。 而我們身為醫師, 我們對於開發個人化的藥物 特別感興趣。 意思就是針對個別癌症病人 設計的療程和免疫治療, 因為癌症治療的難題挑戰之一, 就是每位病人的腫瘤都不一樣。 如果把兩位得到同樣腫瘤的病人 拿來做比較, 兩人腫瘤的相似性不到 3%, 其餘 97% 都非常獨特。 現今,仍然沒有辦法 處理病患腫瘤的獨特性。 因此,我們在尋找能夠用在 免疫治療上的技術, 能夠在最短的時間內 開發出治療方法。 我們的想法是要取得腫瘤的基因序列, 並製造出個人化的疫苗, 且在幾週內就完成製造。
CA: Is it fair to say that almost all of the significant things that happen to us biologically are actions done by proteins, and that it's mRNA that actually makes those proteins? If you can understand the language of mRNA, you can get involved in pretty much everything of significance to the well-being of a human being.
克:是不是可以說, 我們身上發生的各種重要 生物現象,幾乎都是 蛋白質的作為? 而且那些蛋白質是由 mRNA 製造的嗎? 如果你能了解 mRNA 的語言, 你就可以介入幾乎所有 對人類健康很重要的因素?
ÖT: Exactly. So in principle, the information instructions are in the DNA. These have to be translated into protein because proteins are the actors which keep our cells alive and our organism functional. And the way how to translate what is instructed by DNA in a fashion that it is well-timed and happens at the right places, into protein, there is messenger RNA. Messenger RNA sort of instructs when and how much of which protein has to be built in order to ensure the activity of our body.
圖:沒錯。所以,原則上, 訊息的指令位在 DNA 裡面。 它們必須要被轉譯為蛋白質, 因為蛋白質的角色 就是維持細胞的生命 並確保生物組織能運作。 至於要如何轉譯 DNA 的指令, 且要能即時, 要在對的地方發生,進入蛋白質, 那就是 mRNA(信使核糖核酸), 它會指示在什麼時候 要製造多少量的哪一種蛋白質, 才能確保我們的身體能正常活動。
CA: So you can almost think of DNA as the sort of The Oxford English Dictionary of Language. It sort of sits there as the reference point. But for the actual living work, the living work of language out there in the world instructing things, that is done by mRNA.
克:所以可以把 DNA 想成是 語言的牛津字典。 它就在那裡當參考書。 但在實際生活上, 那些活的語言工作, 要下指令的事,就由 mRNA 來做。
UŞ: Yeah, absolutely, it is possible. So the human cells, exactly, DNA is like a library. If you have the platform for the messenger RNA therapy, you can deliver any type of message and the body cells ensure that the message is translated into the right protein.
沙:的確,是有可能的。 人類細胞, DNA 就像是圖書館。 如果有一個 mRNA 治療的平台, 你就可以傳遞任何類型的訊息, 身體細胞會確保那些訊息 被轉譯成正確的蛋白質。
ÖT: A high advantage of mRNA is that it is so versatile. You can deliver all sorts of messages, as Uğur has called them. On the one hand, you can deliver the blueprint for the protein which you want to be produced in this cell. But you can, with the same molecule, also design into the mRNA instructions how this protein should be built, instructions to the protein factories of the cell. So you can define whether you want this protein to be built in high amounts or for a long duration, how the pharmacokinetics of this protein should be in the cell.
圖:mRNA 的一大優勢在於 它的功能非常多。 你可以像沙忻說的那樣 傳遞各種訊息。 一方面,你可以傳遞蛋白質的藍圖, 即你想要在這個細胞中 製造什麼蛋白質。 但是同樣用這個分子, 你也可以將 mRNA 設計成 指示這種蛋白質應如何製造, 指示細胞中的蛋白質工廠。 所以你可以定義 你是否想要大量生產這種蛋白質 或是要長期製造, 這種蛋白質在細胞中 該有怎樣的藥物動力學。
CA: So talk about January of last year when you first heard about this new virus that was spreading.
克:談談去年一月, 當你第一次聽到 這種新病毒在傳播的情況。
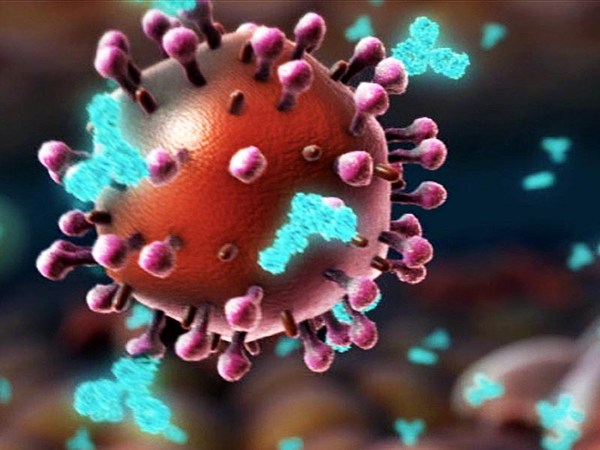
UŞ: So in the end of January, we read a paper published about this outbreak in Wuhan, and realized that this new outbreak has all features to become a global pandemic, and we were concerned that our life will change, that this outbreak could change the fate of mankind. And we knew that we have this messenger RNA technology, which was actually developed for personalized cancer therapy. But the idea of personalized cancer therapy is to get the genetic information of the patient and then make a vaccine as fast as possible. And we had now the same situation. It was not a personalized vaccine, but it was a genetic information of the virus, which was released two weeks earlier. And so this genetic information of this virus was available, and our task was to make a vaccine as fast as possible. And the challenge at that time point was, there was almost nothing known about this virus. It was a completely new virus. We had some assumptions which target which molecule encoded by the virus could be the right target. That means the molecule which can be used to precisely engineer an immune attack. This is the spike protein. It is on the surface of the virus. And there's not only one copy of the spike protein on the virus, but multiple in the range of 20, 25, 30 spike proteins. And the spike protein has two functions. One function is really to enable that the virus sticks to human cells. For example, it sticks to cells in the human lung. And the second is that the spike protein acts as a key. It allows the virus to enter into the cells. Our goal was to engineer an immune response.
吳:去年一月底, 我們讀到一篇論文在談武漢疫情爆發, 意識到這場新爆發的疫情, 具備會成為全球大流行的所有特點, 我們很擔憂我們的生活會起變化, 這場疫情可能會改變人類的命運。 那時我們知道 我們有這種 mRNA 科技, 實際用於開發個人化的癌症治療。 但是個人化的癌症療法 是得到病人的遺傳資訊, 然後在最短的時間內製造出疫苗。 我們現在有同樣的情況。 不是個人化的疫苗, 而是用病毒的遺傳資訊, 這個資訊那之前兩週已經公布了。 所以這種病毒的遺傳資訊已經到手, 我們的任務就是盡快把疫苗做出來。 當時的挑戰是 我們對此種病毒一無所知。 這是種全新的病毒。 我們有一些假設, 猜想這個病毒編碼的那種分子 可能是正確的目標。 這也意味著這個分子可以拿來 精確設計免疫攻擊。 這是棘蛋白。 存在於病毒的表面上。 病毒上的棘蛋白不只一個, 而是落在 20、25、30 個的範圍內。 棘蛋白有兩個功能。 第一個功能是讓病毒 黏附在人體細胞上。 例如,它黏在人類的肺部細胞上。 第二個功能是棘蛋白就像把鑰匙。 它讓病毒能進入細胞內。 我們的目標是設計一種免疫反應。
CA: You've got a slide showing the T-cell response to your vaccine. How long were you into the process before you saw this and you saw, wow, there really is a spectacular response going on here?
克:你有一張圖顯示 T 細胞 對疫苗的反應。 你開始之後等了多久才看到這個? 你看到了,哇,真的有驚人的反應?
ÖT: We saw this already in the animal models because they are also meant to assess the immune response. And what is shown on this slide is on the left side, a lymph node from a setting where there was no RNA treatment or RNA vaccination. And on the right side, a lymph node of a treated organism, in this case, an animal. And the localization matters. And we have constructed our RNA nanoparticles, with encapsulation into lipids such that the mRNA is carried into lymph nodes, not just anywhere, it's carried into lymph nodes and in the lymph nodes it reaches a very special cell type, which is called dendritic cells, and these cells are coaches of the immune system. So they are the generals which call all the different special forces and train them on the wanted poster of attacker. And it's very important that you reach those cells. On the right side, you can see the effect of reaching those cells. You see many red dots. And these are T-cells which have been trained to recognize the antigen, the protein which mRNA has delivered, and they have expanded to a sort of army of clones, so to say. So all these red dots are an army which only knows one goal, namely attacking this specific protein encoded by the mRNA.
圖:我們早就在動物上看到反應, 因為它們也旨在評估免疫反應。 這張圖上顯示 左邊的淋巴結 處在沒有 RNA 治療 或 RNA 疫苗的環境下, 右邊的則是受過治療的 某種生物的淋巴結, 這個例子是一隻動物。 定位也很重要。 我們建構了 RNA 奈米粒子, 以脂質包囊, 讓 mRNA 被帶入淋巴結, 不是到處亂跑,而是被帶入淋巴結, 在淋巴結, 它可接觸到一種非常特別的細胞, 稱為樹突狀細胞, 這些細胞可以指導免疫系統。 所以它們就是將領, 召喚各種特種部隊, 訓練它們找出通緝令上的攻擊者。 能夠傳遞到這種細胞非常重要。 在右邊你們可以看到 傳遞到這些細胞後的效果。 你們可以看到很多紅點。 這些就是 T 細胞, 它們受過訓練、能辨認抗原, 即 mRNA 傳遞的蛋白質, 我們還可以這麼說, 它們會擴充成一支複製人軍隊。 這些紅點就是一支軍隊, 它們只有一個目標, 就是攻擊這個由 mRNA 編碼出的特定蛋白質。
CA: So it's really stunning that within just a few days of your looking at this sequence of the most dangerous pathogen to hit humanity in 100 years, I guess, that you were able to come up with these these candidate vaccines. And I guess over the course of the next weeks and months, you had growing confidence that, wow, this was going to work. It wasn't until the results of the human trials came out, I guess in November of last year, that you really knew. Tell us about that moment.
克:所以這真的很驚人, 在你們看到這個病原體的 基因序列後幾天內, 我猜這百年內打擊人類 最危險的病原體, 就能找出這幾種候選疫苗。 我猜在接下來幾週及幾個月內, 你愈來愈有信心,說哇,這個會有用。 直到人體試驗結果出爐, 我猜是去年十一月, 你才真的確知。 談談那個瞬間。
ÖT: It was a Sunday when we were waiting for these results, which are assessed in such trials by an independent committee and Uğur said, "So let's see how the data will look like." It was not clear whether it would be a thumbs up or down. And we were very relieved. And I felt blessed to hear that the vaccine was efficacious and it was highly efficacious, over 90 percent.
圖:那是個星期日,我們都在等結果。 此類試驗通常都由一個 獨立委員會評估。 沙忻說:「我們來看看 數據會是什麼樣子。」 當時還不清楚會是通過還是否決。 我們都大大鬆了一口氣。 我很欣喜聽到疫苗是有效的, 而且是非常有效,超過 90% 。
CA: And that more than 90 percent almost disguises the full extent, because that's just against any kind of level of infection of COVID. Severe infection and fatalities were almost completely protected against, I think. And it must have been an ecstatic moment for you. Certainly was for so many people around the world.
克:而且那個「超過 90% 」 幾乎涵蓋了所有範圍, 因為可以保護任何程度的新冠感染。 幾乎可以完全保護 不受嚴重感染和死亡, 我想。 你當時一定欣喜若狂。 對世界各地的許多人而言的確如此。
UŞ: Yes, absolutely. So this was a Sunday evening, and there were a handful of people knowing that an effective vaccine is existing against this global pandemic. And we were so excited and so happy and we shared of course this information the next day.
吳:是,絕對是。 那是星期天晚上, 有一小搓人 知道有一種有效的疫苗存在, 可以對抗這個全球疫情。 我們超興奮、超開心, 而且我們隔天就分享了這個資訊。
CA: So based on what's happened this time around and the amazing acceleration, compared with any other vaccine development, I mean, if we were hit by another virus, could you picture that next time we could accelerate the time line further still if need be?
克:所以根據這次發生的事 及驚人的加速度, 跟其他疫苗的開發相比, 如果我們又碰到另一種病毒, 你能想像下次 我們還能更快嗎?如果有需要?
UŞ: Yes, Chris, this is an excellent question. Actually, the world was not prepared to deal with such a pandemic. The science and the vaccine developers reacted in an excellent fashion. And it is incredible and wonderful that it was possible to come up with an effective vaccine while a pandemic is ongoing, in less than 12 months. But the challenges that we have at the moment is that we don't have sufficient production capacity. Ideally, we would be prepared the next time, not only to develop a vaccine in light speed, but also to to make and distribute the vaccine in light speed. So what we need now is an additional element which was not existing, is manufacturing capacity. And idle manufacturing capacity. We must be bringing us into a position that we can produce 12 billion doses of vaccine, if you consider prime boost, within less than six months. And this is technically possible. So this can be done if governments and international organizations invest into manufacturing capacity, invest into keeping this idle capacity, and also come up with a standard time span and process to enable even faster response. So we in principle, we might be able to manage to come up with a vaccine and start distribution in even less than eight months.
吳:是的,克里斯, 這個問題問的太好了。 其實,世界並沒有準備好 應對這樣的疫情。 科學和疫苗開發者以極佳的方式應對, 這的確很不可思議,也很棒, 能找出非常有效的疫苗, 在疫情還在持續發展 不到 12 個月內。 但是我們目前的挑戰 是沒有足夠的生產能力。 理想的情況是,我們應該會 準備好下一次, 不但要以光速發展疫苗, 還要以光速製造及分配疫苗。 所以我們現在需要的 是之前沒有的另一個元素, 就是製造能力。 及閒置產能。 我們必須讓自己達到一個情況, 能生產 120 億劑疫苗, 如果你想在短短六個月內 做初免加強接種方案。 這在技術上是可行的。 要做到這點,政府 及國際組織要投資生產能力, 投資閒置產能, 也要提出標準時限及過程, 才能夠更快地應對。 所以原則上 我們可能可以設法找出一種疫苗 並在八個月內開始分配。
CA: What does what's happened in this last year tell you now about the prospects for using mRNA to treat cancer and indeed other diseases? Where is this heading?
克: 從現在看去年發生的事, 使用 mRNA 治療癌症 甚至其他疾病的前景如何? 未來的發展方向?
UŞ: What we have now is now an approved technology and a first approved product. The development of the coronavirus mRNA vaccine shows the power of the mRNA and it shows also the safety of this approach. And it shows that it opens up a door for new technology and for new type of treatments. And the mRNA molecules that we are currently using for cancer, we have more than 10 products now in clinical development, are diverse against different types of cancer. We are very confident that the success that we have generated now for our infectious disease vaccines can be continued with our cancer immunotherapies.
吳:我們現在有已經核准的科技 以及第一個核准的產品。 新冠病毒 mRNA 疫苗的發展顯示出 mRNA 的力量, 也顯示出這個方法的安全性。 它指出了 一扇新科技 及新療法的門。 mRNA 分子 我們目前用來治療癌症的, 現在我們有超過十種產品 正在做臨床開發, 可以對抗不同類型的癌症。 我們非常有信心目前 對抗傳染病疫苗的成功經驗, 可以持續到癌症的免疫療法。
CA: Some people may hear this and say this is just another type of drug that's coming along. But I think on the mental model you're talking about, we should think about it as much more revolutionary than that, that typically a drug, a traditional drug, kind of changes the chemical environment, the background of an entire area of the body. But your -- If you understand the language of mRNA, you can do something much more specific and precise. Is that something like a fair way to think about it?
克:有些人聽到這個, 大概會說這只不過是另一種新的藥物。 但是我認為從你談到的 心智模式角度來看, 我們應該認為這更具革命性, 比普通藥物、傳統藥物更加革命性, 可以改變化學環境, 即整個人體的背景狀況。 但是你的—— 如果你了解 mRNA 的語言, 你就可以做得更具特異性、更精準。 這樣說公平嗎?
ÖT: Yes, indeed. It could be the next revolution in the biopharm landscape.
圖:是,確實。 這很可能是 生物製藥領域的下一場革命。
UŞ: At the end of the day, disease is a situation where the communication between cells is disturbed. So, for example, autoimmune disease is a disease condition where immune cells attack normal cells. And indeed, we could engineer messenger RNA therapies which could teach the immune system to stop to do that, without inhibiting the whole immune system, by just communicating with the immune cells which are attacking. We could be precise and more specific.
吳:到頭來,疾病不過就是 細胞間溝通不良的情況。 例如自體免疫疾病就是 免疫細胞攻擊正常細胞的狀況。 確實,我們可以編碼信使 RNA 療法, 教免疫系統停止這樣做, 不需要抑制整個免疫系統, 只要直接傳達給攻擊的免疫細胞就好。 我們可以更精確及更具特異性。
CA: The success of BioNTech over the last couple of years, I think the value of the company has rocketed because of the amazingness of what's happened. I mean, it's made you both extremely wealthy, I think you're both billionaires now. How have you been able to respond to that? Sometimes so much money brings its own problems with it. Is that proving a distraction?
克:生技公司 BioNTech 近幾年來的成功, 我想公司的價值飛漲, 因為發生的這一切太神奇了。 這讓你們兩位非常富有, 我想你們兩位現在都是億萬富翁了。 你們對此有何反應? 有時候太多錢本身也會帶來問題。 這證明會讓人分心嗎?
ÖT: For a company which sees innovation as its core mission, too much money is never a problem. Because innovation really means that you have to invest. Otherwise, we will only have two type of products or incremental improvement for solutions of high medical need.
圖:對一家 視創新為核心使命的公司而言, 錢太多永遠不是問題。 因為創新真正的意思就是投資。 不然我們將只有兩種產品, 或只能漸進式改良 高醫療需求的解方。
UŞ: It really gives us the chance to transform our company. So we were when we started -- When we compare ourselves with the situation we had at the beginning of 2020, we had a number of product candidates in clinical testing, but the company required funding every year or every second year. Now we have a situation to really address the full vision of the company. We started BioNTech with the idea really to provide novel treatments wherever there is a high unmet medical need. And we now can do that in a much larger and broader scale, and bring our innovations faster to patients.
吳:它真的讓我們有機會 改造我們的公司。 我們剛開始的時候—— 如果我們跟 2020 初的景況比較, 我們有幾個臨床試驗的候選產品, 但是公司每年或每隔一年都要找錢。 現在我們終於 能說出公司的完整願景。 我們開創 BioNTech 抱持的想法就是提供新治療方法 給還未滿足的高醫療需求。 現在我們可以用更大 更廣的規模來做, 更快地將我們的創新帶給病患。
CA: You are both from families who immigrated from Turkey to Germany. Immigrants have faced hard times in many countries, including Germany. And yet you, I think, have helped transform the debate about immigration, in Germany and elsewhere, just by the extraordinary success that you've achieved creating this world-leading company in Germany. Do you take joy for the impact you may have had on this issue?
克:你們兩位都來自從土耳其 移民到德國的移民家庭。 移民的處境在許多國家都很艱難, 德國也包括在內。 然而我想,你們 幫助轉變對移民的爭論, 無論在德國及其他地方, 因為你們達到的非凡成就, 在德國創立領導世界的公司。 也許你對這個議題已經產生衝擊, 你為此感到高興嗎?
UŞ: It is somehow surprising because the way how we do science, and how we recognize how people work effectively in teams together is not to us from where the person is coming, but what the person can contribute. So in our company, we have employees from more than 60 countries. So we are an international group of scientists, as any other research institution. So we have to recognize that globalization really helps to bring people, scientists or other engineers into one place, allowing to work together and to come with extraordinary results. For us, this is somehow surprising that this is seen as special. It is just the way how excellent research and science work.
吳:這有點令人驚訝, 因為我們做科學的方法, 以及我們如何認定人們在團隊內 是否能合作無間的方法, 不是你從哪裡來, 而是這個人能貢獻什麼。 所以在我們的公司, 我們的員工從六十多個國家來。 我們是一群來自世界各地的科學家, 就像任何其他研究單位一樣。 所以我們必須明瞭全球化的確有助於 將人、科學家或其他工程師 帶到一個地方, 讓他們能一起工作 並產生非凡的結果。 我們很驚訝這被視為特殊情況。 這其實就是卓越的研究 及科學的運作方式。
CA: Well, it's extraordinary and inspiring what you've achieved, and it'll be very exciting to track progress over the coming years. Thank you so much. Thank you.
克:你們的成就極非凡、極激勵人心, 非常興奮能繼續追蹤未來幾年的進展。 真的很感謝。謝謝。